Inappropriate prescription of corticosteroid therapy during inflammatory ileo‐colitis revealing disseminated tuberculosis with digestive involvement: Two case reports - Razafindrazoto - 2021 - Clinical Case Reports - Wiley Online Library
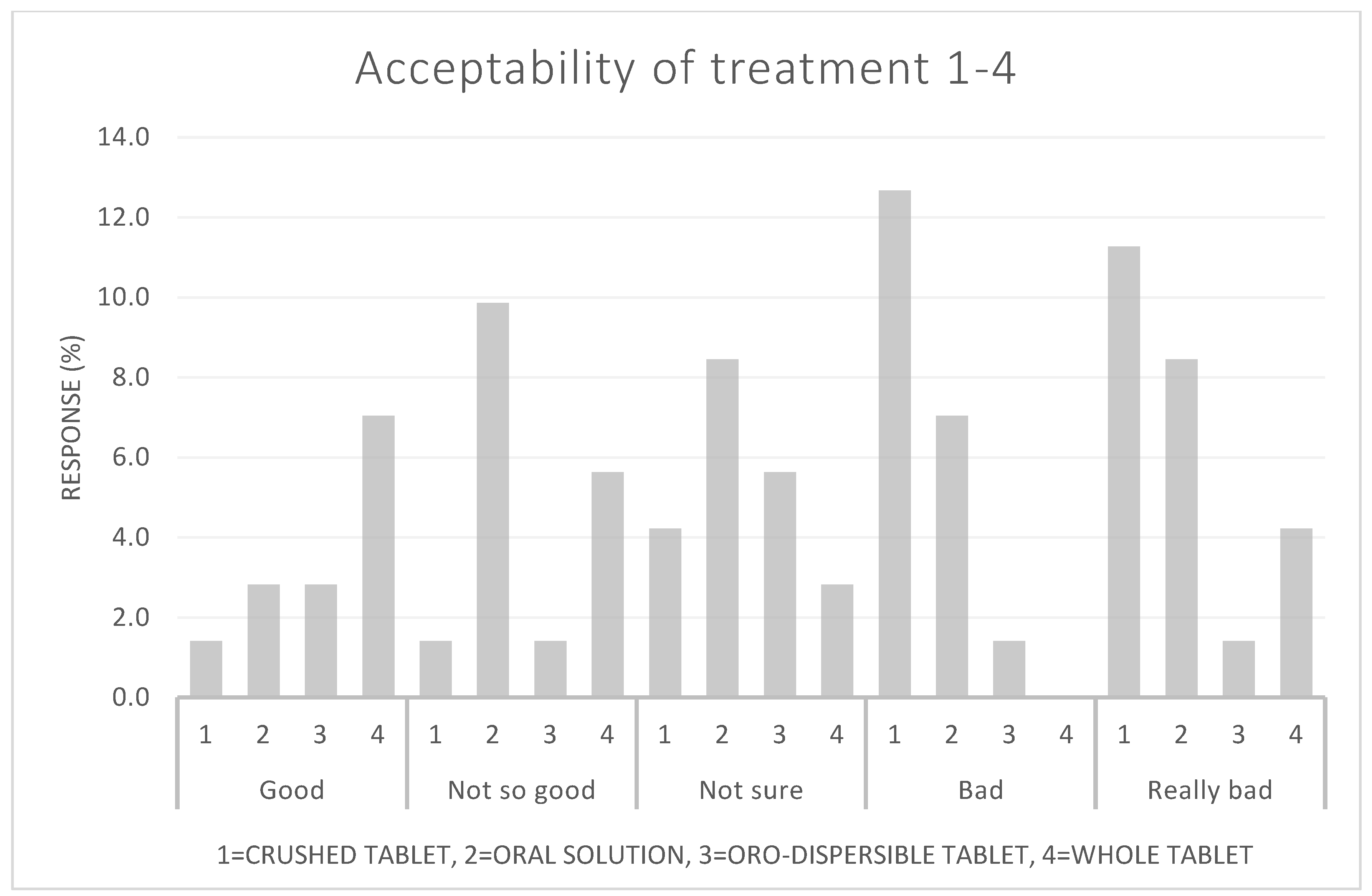
Children | Free Full-Text | Acceptability of Prednisolone in an Open-Label Randomised Cross-Over Study—Focus on Formulation in Children

In Vitro Antioxidant, Anti-inflammatory, and In Vivo Anticolitis Effects of Combretin A and Combretin B on Dextran Sodium Sulfate-Induced Ulcerative Colitis in Mice
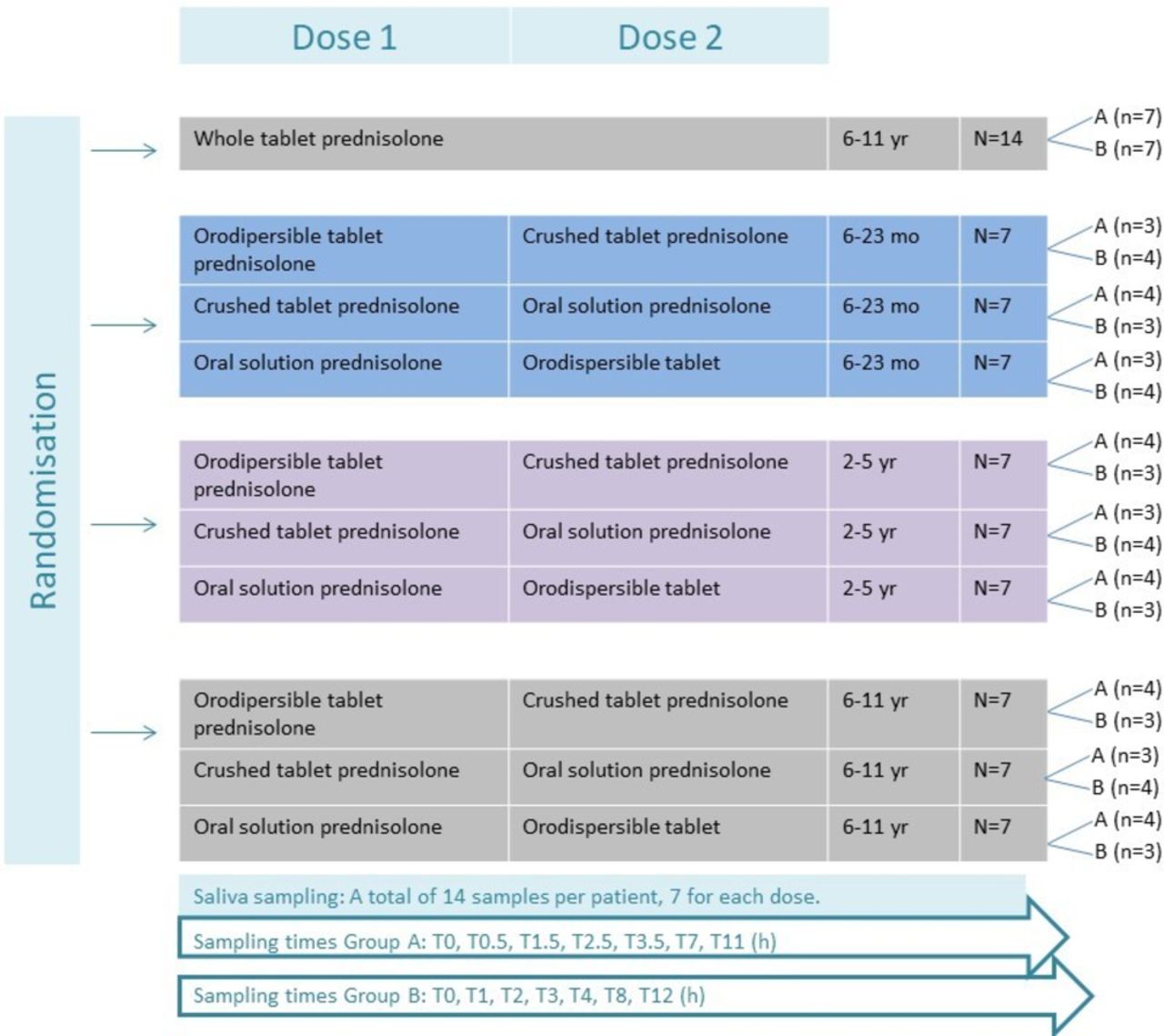
Pharmacokinetics of prednisolone in children: an open-label, randomised, two-treatment cross-over trial investigating the bioequivalence of different prednisolone formulations in children with airway disease | BMJ Paediatrics Open